PART IV — FAMILY II: Halictidae (Sweat Bees): Ecology, Evolution, Social Behaviour and Adaptability
- Dawid Cierniewski
- Apr 23
- 40 min read
21. Introduction to Halictidae: Iridescence, Adaptability, and a Different Kind of Success

If the Andrenidae are defined by subtlety, then the Halictidae are defined by contrast - not only in appearance, but in behaviour, ecology, and evolutionary trajectory. Where mining bees often pass unnoticed, halictids tend to interrupt perception: a flash of metallic green under sunlight, a rapid, almost erratic flight path, or the curious moment when a small bee lands briefly on human skin. These are the so-called “sweat bees,” a name derived from their attraction to salts in perspiration.
Yet, as with many common names in natural history, the label captures a behaviour, not a system - and in doing so, obscures far more than it reveals.
The Halictidae comprise over 4,000 described species globally, with estimates suggesting substantially higher undescribed diversity, particularly in tropical regions (Michener, 2007; Ascher & Pickering, 2020). They are among the most ecologically dominant wild bee families, especially in temperate and semi-arid systems. Large-scale community studies consistently show that halictids - particularly Lasioglossum and Halictus - can account for 20–60% of local bee abundance, particularly in disturbed or anthropogenically modified habitats (Winfree et al., 2011; Roulston & Goodell, 2011).
This numerical dominance is not incidental. It reflects a set of biological properties that allow halictids to persist - and often thrive - under conditions where more specialised taxa decline.
At first glance, this success might appear to stem from diversity alone. But diversity, in itself, explains very little. The more interesting question - a question that has long occupied naturalists and evolutionary biologists alike - is how such diversity is structured and maintained. Early interpretations of insect societies often treated social behaviour as an endpoint of evolutionary progression, a movement from simplicity toward complexity. Yet Halictidae complicate this view.
Within this family, complexity does not accumulate in a single direction. Instead, it appears, disappears, and re-emerges under different conditions. Eusociality is not fixed, but contingent; solitary behaviour is not primitive, but persistent. What initially looks like variation becomes something more revealing: a system in which multiple strategies are continuously tested against the environment, retained when advantageous and discarded when not.
In this sense, halictid diversity is not simply taxonomic - it is functional, reflecting an ongoing interplay between ecological constraint and evolutionary possibility.
What distinguishes the family is not simply the number of species it contains, but the range of strategies expressed within it.
Unlike Andrenidae, which are overwhelmingly solitary, halictids span the full spectrum of social organisation: solitary, communal, primitively eusocial, and facultatively eusocial systems all occur within closely related taxa. Molecular phylogenetic analyses indicate that eusociality has evolved multiple times independently within the family - at least three to four origins - with several reversals back to solitary behaviour (Danforth et al., 2006; Brady et al., 2006; Gibbs et al., 2012).
This is an extraordinary pattern. In most animal systems, complex social behaviour, once evolved, tends to persist. In Halictidae, it does not. It appears, disappears, and reappears - suggesting that sociality is not a fixed endpoint, but a repeated evolutionary experiment.
At this point, it is worth pausing, because this challenges a persistent intuition in biology: that evolution tends toward increasing complexity. Halictidae undermine that view. Phylogenetic evidence shows that eusociality has evolved multiple times within the family, but has also been repeatedly lost when conditions no longer favour cooperation (Brady et al., 2006; Gibbs et al., 2012; Field et al., 2010).
This demonstrates a simple but often overlooked principle: natural selection does not favour complexity - it favours fitness. When cooperative systems increase reproductive success, they are maintained; when they do not, they are lost.
In this sense, the loss of sociality is not regression. It is adaptation.
Evolution does not move toward complexity - it moves toward whatever works.
Before reaching that behavioural complexity, however, the most immediate point of contrast is visual.
Many halictid bees exhibit striking metallic colouration - greens, blues, bronzes, and copper tones - that differ fundamentally from pigment-based colour systems. Rather than relying solely on chemical absorption, these colours are produced through structural colouration, generated by nanoscale multilayer arrangements within the cuticle that create interference effects (Kinoshita, 2008; Seago et al., 2009).
In practical terms, this means the colour is not “applied” to the body, but emerges from its structure. The bee is not green in the way a leaf is green; it is green in the way oil on water appears iridescent - because light itself is being manipulated.
This distinction is more than aesthetic. It reflects a broader theme in halictid biology: function emerging from structure across multiple scales. The same principle that governs nanoscale light interference reappears at the ecological level in the form of behavioural flexibility.
Importantly, metallic colouration is not universal within the family. Many halictids - particularly within subgenera such as Dialictus - are matte black or weakly lustrous. This variation reinforces a critical point: Halictidae are not defined by any single morphological trait. Instead, they are characterised by a range of possible forms built upon a shared structural framework.
The same pattern holds for nesting behaviour.
Like Andrenidae, many halictids are ground-nesting, excavating burrows and provisioning brood cells with pollen and nectar. However, where mining bees tend to operate as strictly independent units, halictid nests can become sites of interaction - shared, defended, or cooperatively maintained depending on ecological context. The nest, in this sense, is not just a structure, but a potential social arena.
Their ecological success is closely tied to this capacity for flexibility. Halictidae occupy a wide range of habitats, from undisturbed grasslands to heavily modified urban environments. Empirical studies show that halictids are often disproportionately abundant in disturbed systems, where traits such as small body size, generalist foraging, and flexible nesting behaviour allow them to exploit fragmented and unpredictable resources (Winfree et al., 2007; Potts et al., 2010).
In urban landscapes, for example, Lasioglossum species are frequently recorded nesting in compacted soils, pavements, and garden substrates - conditions that would exclude many specialist ground-nesting bees. This is not simply tolerance; it is functional adaptation to instability.
Their foraging behaviour follows the same pattern. Many halictids are polylectic, visiting a wide range of plant taxa. This generalism acts as a buffer against environmental variability, allowing individuals to maintain resource intake even as floral communities shift. Yet even here, the picture is more complex than it first appears. Within broadly generalist species, individuals often exhibit short-term preferences or “micro-specialisations,” suggesting that flexibility operates at multiple scales simultaneously.
There is also a physiological dimension to their interaction with the environment - one that occasionally brings them into direct contact with us.
The attraction of some halictids to human sweat reflects mineral foraging, particularly for sodium, which is often limiting in nectar and pollen. This behaviour, known as puddling, is widespread across insects and represents a strategy for balancing nutrient intake beyond basic energetic requirements (Arms et al., 1974).
It is tempting to dismiss this as a curiosity - an odd behavioural quirk - but it is, in fact, a reminder of something deeper: that organisms are not optimised for single resources, but for nutritional balance across multiple constraints.
From an evolutionary perspective, the Halictidae represent not just a lineage, but a system in which strategies remain unusually flexible. Where Andrenidae demonstrate the long-term success of stability - precision in nesting, timing, and resource use - Halictidae demonstrate the success of variability itself.
They do not replace the framework established by mining bees; they expand it.
They show that evolutionary success in bees can be achieved in more than one way: through consistency, but also through responsiveness; through specialisation, but also through generalism; through independence, but also through cooperation when conditions allow.
And it is precisely this combination - structural diversity, behavioural plasticity, and ecological adaptability - that makes Halictidae not just another bee family, but one of the most important systems for understanding how evolution operates under changing conditions.
21.1. Defining Halictidae: Morphological Identity and Taxonomic Boundaries
While much of the ecological and behavioural interest surrounding Halictidae lies in their flexibility, the family itself is defined by a suite of morphological characters that distinguish it from other bee lineages. These traits are often subtle and not immediately apparent in the field, but they provide the structural basis for taxonomic classification and reflect a shared evolutionary history (Michener, 2007; Danforth et al., 2006).
As in most bees, identification relies on combinations of characters rather than a single defining feature. In Halictidae, one of the most important diagnostic regions is the wing. The forewing venation typically shows a strongly arched basal vein and a characteristic arrangement of submarginal cells, features that distinguish halictids from closely related families such as Andrenidae and Colletidae (Michener, 2007). Although these differences may appear minor, wing venation patterns are highly conserved across bee lineages and are widely used in higher-level systematics.
Mouthpart morphology provides a second key distinction. Halictidae belong to the short-tongued bee assemblage (traditionally Anthophila: ‘non-long-tongued’ bees), characterised by relatively short glossae compared to long-tongued groups such as Apidae and Megachilidae (Michener, 2007; Danforth et al., 2019). Within this assemblage, however, halictids differ from families such as Colletidae in details of the labium and maxillary structures, reflecting differences in nectar uptake and processing mechanisms. These features are not tied to a single floral type, but instead support the broader generalist tendencies observed in many species.
Pollen transport structures further contribute to their identification. Most female halictids carry pollen on the hind legs using scopal hairs, a trait shared with Andrenidae, but typically less specialised in structure and density than in oligolectic taxa (Michener, 2007; Westrich, 1996). This relatively unspecialised scopal morphology is consistent with the predominance of polylectic foraging within the family.
Additional diagnostic characters include features of the thoracic and abdominal morphology. The pronotum is typically short and does not extend back to the tegula, distinguishing halictids from some other bee families, while females often possess a well-developed pygidial plate used in nest construction (Michener, 2007). Many of these traits require microscopic examination, emphasising that taxonomic identity is determined by fine-scale structural features rather than gross external appearance.
Importantly, these morphological criteria are now strongly supported by molecular phylogenetics. DNA-based analyses consistently recover Halictidae as a monophyletic lineage within the short-tongued bees, reinforcing classifications based on morphology despite the extensive ecological and behavioural variation observed within the group (Danforth et al., 2006; Cardinal & Danforth, 2013).
What follows from this is a critical distinction. The features that define Halictidae are structural and phylogenetic, not ecological. Metallic colouration, attraction to sweat, and flexible social organisation - while characteristic of many species - are not diagnostic of the family. They are variable expressions within a lineage that is, at its foundation, morphologically coherent.
And it is precisely this combination that makes Halictidae so informative. Once the taxonomic boundaries are fixed, the diversity of behaviours observed within the family can be understood not as differences between unrelated organisms, but as alternative strategies explored within a single evolutionary framework.
22. Evolutionary Position and the Emergence of Social Flexibility
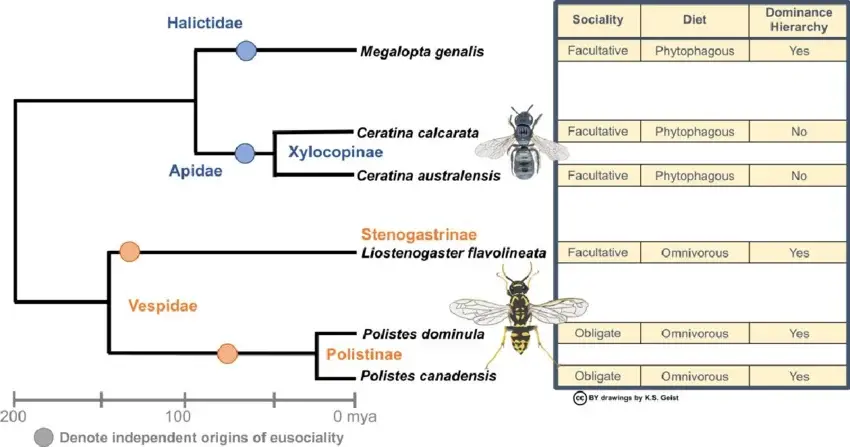
Within the phylogeny of bees, the Halictidae occupy a position that is not exceptional in terms of lineage placement, but is unusually informative in terms of evolutionary process. As part of the short-tongued bee assemblage (Melittidae, Andrenidae, Halictidae, Colletidae), they form a well-supported clade within Anthophila (Danforth et al., 2006; Cardinal & Danforth, 2013). However, while closely related families such as Andrenidae exhibit relatively conserved life-history strategies - predominantly solitary nesting with limited behavioural divergence - the Halictidae are characterised by repeated transitions in social organisation, making them one of the most important empirical systems for understanding the evolution of eusociality.
Phylogenetic analyses based on molecular data indicate that eusocial behaviour has evolved multiple times independently within Halictidae, with estimates suggesting at least three to four separate origins, alongside several independent reversals to solitary behaviour (Brady et al., 2006; Gibbs et al., 2012). This pattern contrasts sharply with that observed in highly eusocial lineages such as Apidae, where social organisation appears to have evolved once and subsequently stabilised through morphological and behavioural canalisation. The halictid pattern is therefore not simply one of diversity, but of evolutionary lability. Sociality is gained, modified, and lost within relatively short evolutionary timescales, indicating that it is neither a fixed endpoint nor an irreversible trajectory.
This observation challenges a long-standing assumption in evolutionary biology: that increasing social complexity represents a unidirectional progression. Classical interpretations, influenced in part by early theoretical frameworks such as those developed by William D. Hamilton (1964), emphasised genetic relatedness - particularly under haplodiploid sex determination - as a primary driver of eusocial evolution. While kin selection theory remains fundamental, the halictid system demonstrates that genetic predisposition alone is insufficient to explain observed patterns. If relatedness were the dominant determinant, one would expect social organisation to be relatively stable across populations. Instead, closely related populations - and in some cases, populations of the same species - exhibit markedly different social structures under different environmental conditions (Gibbs et al., 2012).
The importance of ecological context is most clearly illustrated in studies of Halictus rubicundus, which exhibits facultative eusociality across its geographic range. In regions characterised by longer growing seasons (typically exceeding 12–16 weeks), colonies display eusocial organisation, with an initial brood functioning as workers and contributing to the production of a second reproductive brood. In contrast, populations in shorter-season environments (approximately 6–8 weeks) remain solitary, producing only a single brood with no worker phase (Eickwort et al., 1996; Field et al., 2010). This transition is not gradual but threshold-dependent, reflecting the temporal constraints imposed by development and resource availability.
The underlying mechanism is closely tied to life-history scheduling. Eusociality in this system requires a sequential reproductive strategy in which early offspring forego direct reproduction in order to assist in raising subsequent brood. The fitness advantage of this strategy depends on whether the additional reproductive output generated by cooperative behaviour exceeds the initial cost of worker production. Where season length permits two or more reproductive cycles, this condition can be met; where it does not, solitary reproduction remains the optimal strategy (Seger, 1983; Field et al., 2010). In this sense, eusociality can be understood as a conditional investment strategy, with its viability determined by the interaction between developmental timing and environmental duration.
Such findings have broader implications for how social evolution is conceptualised. Rather than representing discrete categories, the social systems observed in Halictidae form a continuum, ranging from solitary nesting through communal associations to primitively eusocial and facultatively eusocial organisations (Wilson, 1971; Bourke, 2011). Importantly, these are not merely descriptive classifications but reflect underlying differences in reproductive integration, division of labour, and temporal organisation. In communal systems, for example, multiple females may share a nest entrance but retain independent reproductive roles, whereas in primitively eusocial systems, reproductive skew emerges, albeit without fixed morphological caste differentiation. This gradation provides a rare opportunity to examine intermediate stages in social evolution directly, rather than inferring them from comparisons between distantly related taxa.
However, it is equally important to recognise that the presence of intermediate states does not necessarily imply a linear evolutionary pathway. The repeated emergence and loss of eusociality within Halictidae suggests that these states are not steps along a fixed trajectory, but alternative solutions to the same ecological problem: how to maximise reproductive success under varying environmental constraints. In this framework, solitary and social strategies are not hierarchically ordered, but context-dependent, each favoured under different combinations of resource availability, season length, and environmental stability.
The reversibility of social organisation within Halictidae further reinforces this interpretation. Transitions from eusocial to solitary behaviour - documented both phylogenetically and in contemporary populations - indicate that the costs of sociality can outweigh its benefits under certain conditions (Gibbs et al., 2012). These costs include reproductive suppression of workers, increased competition within nests, and elevated exposure to parasites and pathogens due to group living. Where environmental conditions reduce the potential benefits of cooperation - particularly through shortened seasons or reduced resource predictability - selection can favour a return to solitary reproduction. This demonstrates that eusociality, while often treated as a pinnacle of social evolution, is in fact a contingent and potentially unstable strategy.
Taken together, the evolutionary dynamics of Halictidae suggest that the emergence of complex social behaviour may be more accessible than previously assumed, but also more fragile. Eusociality does not require highly specialised or rare conditions to arise, but neither is it guaranteed to persist. Instead, it occupies a region of adaptive space defined by a balance between ecological opportunity and constraint.
In this sense, the position of Halictidae within bee phylogeny is best understood not in static terms, but as a dynamic system in which behavioural strategies are continually tested against environmental conditions. Their significance lies not only in their diversity, but in their capacity to reveal social evolution as an ongoing, reversible process. By retaining the ability to move between solitary and social states, halictid bees expose the conditional nature of complexity itself, demonstrating that even highly organised systems are ultimately shaped - and limited - by the ecological contexts in which they operate.
23. Morphology and Colouration: The Physics of Metallic Bees

Few encounters in bee biology are as immediately striking as the sight of a metallic halictid under direct sunlight. Greens shift into bronze, blue flashes appear and disappear, and the bee seems almost to change substance as it moves. It is tempting to interpret this as ornament - a decorative layer over an otherwise ordinary form. But that reading is misleading. In halictids, colour is not simply appearance; it is a consequence of structure, and structure in biology is rarely incidental.
Most animal colouration arises through pigments, where molecular structure determines which wavelengths of light are absorbed and which are reflected. Structural colouration operates through a different mechanism. Here, colour is generated by the interaction of light with nanoscale architecture, through processes such as interference, scattering, and diffraction. In metallic halictids, this is typically achieved through multilayer reflectors within the cuticle: thin layers with differing refractive indices that partially reflect incoming light, producing interference patterns that amplify specific wavelengths while cancelling others (Kinoshita, 2008; Seago et al., 2009).
The scale of this architecture is critical. Layer thicknesses are typically on the order of ~100–300 nm, meaning that even minor variation can produce substantial shifts in reflected wavelength. This explains how closely related species - or even individuals - can exhibit noticeably different colours. The greens and blues commonly observed in halictids correspond to constructive interference in the ~450–550 nm range, which also overlaps with peak sensitivity in bee visual systems (Kinoshita, 2008).
Unlike pigment-based colours, structural colours are often angle-dependent. As viewing angle changes, the optical path length through the layers shifts, altering the wavelengths that are constructively reinforced. The result is iridescence: colour that is dynamic rather than fixed. A halictid may appear bright green from one angle, darker or bronze from another, and nearly black under diffuse light. This instability is not a flaw - it is a defining feature of the mechanism itself (Seago et al., 2009).
Understanding what this colour does biologically is less straightforward.
One hypothesis is that structural colouration contributes to camouflage through dynamic reflectance. In visually complex environments, such as vegetation with dappled light, an unstable colour signal may reduce predator detection by disrupting edge recognition. Rather than blending into a background through static resemblance, iridescent surfaces may evade detection by refusing to present a consistent visual target (Seago et al., 2009).
A second hypothesis involves thermoregulation. Small-bodied bees, including many halictids (typically ~3–12 mm), experience rapid heat exchange with their environment due to high surface-area-to-volume ratios. Structural surfaces may influence how solar radiation is absorbed or reflected, potentially affecting body temperature. Some recent work suggests correlations between structural colour and environmental conditions, but empirical support remains limited and likely context-dependent rather than universal (Kinoshita, 2008).
A third possibility is visual signalling, particularly in short-range interactions such as mate recognition. While bees rely heavily on chemical cues, visual traits may still contribute to species discrimination or behavioural responses. However, current evidence does not support a single, dominant signalling function for metallic colouration across Halictidae, and its role likely varies among taxa.
What emerges from these competing explanations is not a single answer, but an important insight: conspicuous traits do not always map cleanly onto single functions. Structural colour may persist because it contributes weakly to multiple functions, or because it arises as a by-product of cuticular architecture shaped by other selective pressures. In this sense, its evolutionary significance may lie as much in constraint and correlation as in direct adaptation.
Equally important is that metallic colouration is not universal within Halictidae. While genera such as Agapostemon, Augochlora, and some Lasioglossum exhibit strong iridescence, many others - particularly within subgenera such as Dialictus - are matte black or only weakly lustrous. This variation reinforces a key point: Halictidae are not defined by a single visual trait, but by a broader framework of morphological and ecological flexibility (Michener, 2007).
Beyond colour, halictid morphology reflects a distinct ecological strategy. Most species are relatively small, typically ranging from ~3 to 12 mm in length. This size range allows access to a wide variety of floral resources, including small or shallow flowers that may be inaccessible to larger bees. It also facilitates exploitation of spatially fragmented habitats, where resources occur in small, dispersed patches (Michener, 2007).
Their body form is generally compact and agile, supporting rapid, short-distance flight. To an observer, their movement may appear erratic, but functionally it reflects an efficient sampling strategy: brief visits, low handling time, and frequent switching between floral units. This behaviour aligns with generalist foraging strategies, where success depends on maintaining efficiency across variable resource distributions rather than maximising extraction from a single source.
Pollen transport structures further support this flexibility. Like Andrenidae, most halictids carry pollen on the hind legs using scopal hairs, but these structures vary in density and arrangement. In generalist species, scopae are sufficiently versatile to handle pollen from multiple plant taxa, rather than being specialised for a single host. This morphological generality underpins the predominance of polylecty within the family (Westrich, 1996; Michener, 2007).
Sensory systems reinforce this ecological versatility. Halictid bees integrate visual and olfactory cues to locate and assess floral resources, with antennae adapted for detecting volatile compounds and compound eyes supporting navigation and pattern recognition (Hempel de Ibarra et al., 2014). These systems allow rapid behavioural responses to changing environmental conditions, consistent with a flexible foraging strategy.
Taken together, the morphology and colouration of Halictidae illustrate a broader principle: biological complexity does not always arise through extreme specialisation. It can also emerge through the refinement of systems that perform effectively across a wide range of conditions.
The metallic sheen of a halictid bee is therefore not merely aesthetic. It is the visible expression of nanoscale structure interacting with physical laws. More broadly, their morphology reflects a lineage optimised for responsiveness - capable of operating efficiently across heterogeneous environments rather than being narrowly tuned to a single ecological niche.
In this way, Halictidae reveal a different kind of sophistication: one rooted not in rigid specialisation, but in precision within flexibility.
And it is in that interplay between physics, morphology, and ecology that their true significance begins to emerge.
24. Nesting and Social Organisation in Halictidae: A Continuum of Social Strategies
The Halictidae occupy a distinctive position within bee biology, defined not by a single form of social organisation, but by the presence of a continuous range of social strategies expressed within a single lineage. This range extends from strictly solitary nesting through communal associations to primitively eusocial and facultatively eusocial systems. Such breadth is rare among insects and renders the family one of the most informative model systems for examining how social behaviour evolves, persists, and, critically, is lost (Michener, 2007; Brady et al., 2006; Gibbs et al., 2012).
At its foundation, halictid biology retains the ancestral condition of solitary nesting. In this system, a single female excavates a burrow, constructs brood cells, provisions each with a pollen–nectar mass, and seals them independently. Development proceeds without further parental involvement once the cell is closed, and reproductive success is therefore tightly constrained by individual foraging efficiency, time, and environmental stability. In many ways, this represents the baseline from which all other social strategies emerge: a system in which everything depends on the success of a single individual, with no buffering from cooperation and no division of labour to offset failure.
The first departure from this condition appears in communal nesting systems. Here, multiple females utilise a shared nest entrance and, in some cases, portions of a common burrow network, yet each provisions and reproduces independently. There is no reproductive division of labour and no caste differentiation. The system is therefore structurally collective but functionally individual - shared infrastructure without shared reproduction. The benefits are subtle but meaningful: reduced excavation costs, reuse of established tunnels, and potential dilution of predation risk at the nest entrance. Yet these associations remain evolutionarily shallow. Because cooperation does not extend to brood care or reproductive integration, the underlying unit of selection remains the individual female, and the system does not fundamentally alter the dynamics of reproduction (Michener, 2007).
A more substantial transition is observed in primitively eusocial systems, where reproductive roles begin to diverge. In these systems, a foundress female establishes a nest and produces an initial brood, a proportion of which remains as workers. These individuals assist in provisioning and brood care, while the foundress retains primary reproductive status. Unlike highly eusocial insects, caste differentiation is not morphologically fixed. Workers are often only partially suppressed reproductively and may retain the capacity to reproduce under certain conditions. Hierarchies are therefore not rigid, but negotiated - roles can shift, dominance can change, and the system remains responsive to internal and external conditions. What emerges is cooperation without complete commitment: a society that functions collectively, but has not yet surrendered individual flexibility (Wilson, 1971; Michener, 2007).
The defining feature of Halictidae, however, lies in facultative eusociality, where social organisation itself becomes conditional. The same species - and even populations within that species - can express either solitary or eusocial behaviour depending on environmental context. This is most clearly demonstrated in Halictus rubicundus, where populations in regions with extended growing seasons produce worker broods and operate eusocially, while those in shorter-season environments remain solitary, producing only a single brood (Eickwort et al., 1996; Field et al., 2010).
What determines this shift is not genetic difference in any simple sense, but time.
Eusociality requires a sequence: an initial investment phase, in which early offspring function as workers, followed by a reproductive phase that generates additional offspring. The success of this strategy depends on whether the second phase can occur. If the season is long enough, cooperation pays off. If it is not, the system collapses back to solitary reproduction. In practical terms, this creates a threshold - often on the order of weeks - beyond which sociality becomes viable and below which it does not (Seger, 1983; Field et al., 2010).
Seen this way, social organisation is less like a fixed trait and more like a decision shaped by environmental constraints - though it is a decision made not consciously, but through the filtering of evolutionary success across generations.
This environmentally mediated flexibility has significant evolutionary implications. Phylogenetic analyses indicate that eusociality has evolved multiple times within Halictidae and has also been lost repeatedly (Brady et al., 2006; Gibbs et al., 2012). Such repeated gain and loss suggests that social organisation within the family is inherently unstable. Eusociality is not a permanent evolutionary “advance,” but a strategy maintained only while its benefits exceed its costs. These costs include reproductive suppression of workers, increased competition within the nest, and heightened exposure to parasites and pathogens associated with group living. When environmental conditions shift - particularly through reductions in season length or resource predictability - selection can favour a return to solitary behaviour.
This challenges the traditional view of social evolution as a linear progression toward increasing complexity. In Halictidae, the evidence instead supports a reversible system, in which solitary and social strategies represent alternative solutions rather than successive stages. Solitary nesting offers independence and low risk but limited scalability; eusociality offers increased efficiency and reproductive output but introduces dependency and vulnerability. The system moves between these strategies not because one is universally superior, but because the environment determines which is viable at any given time.
This dynamic is also reflected in nest architecture. While all halictids share a broadly similar ground-nesting template, structural differences emerge across the social spectrum. Solitary nests tend to be relatively simple, with sequentially provisioned brood cells and minimal overlap between developmental stages. In contrast, eusocial nests often exhibit increased branching, spatial organisation, and the simultaneous presence of multiple cohorts, enabling continuous provisioning and extended reproductive output. These structural differences are not incidental - they are the physical expression of behavioural strategy, linking social organisation directly to the material structure of the nest.
Taken together, the nesting systems of Halictidae are best understood as a continuum shaped by the interaction between environmental constraint, life-history strategy, and evolutionary history. This continuum does not simply illustrate variation; it reveals process. It allows us to observe, within a single lineage, how cooperation emerges, stabilises, and dissolves.
And perhaps most importantly, it reframes what sociality is.
Not an endpoint. Not a pinnacle.
But a conditional state - one that exists only so long as the environment allows it to.
25. Foraging Ecology and Generalism in Halictidae: Flexibility as a Foundation of Stability
In contrast to the more constrained foraging strategies observed in many Andrenidae, the Halictidae are characterised by a high degree of dietary flexibility, with the majority of species exhibiting polylectic behaviour. Rather than specialising on a narrow range of host plants, halictid bees typically exploit a wide diversity of floral resources, collecting pollen and nectar from multiple plant families across a given season (Michener, 2007; Westrich, 1996). This distinction is not merely descriptive - it reflects a fundamentally different ecological strategy.
Floral resources are inherently uneven, both in space and time. Even in relatively stable ecosystems, flowering is staggered, patchy, and often short-lived. In anthropogenically altered environments, this variability becomes even more pronounced, with plant communities shifting rapidly in composition and abundance. Under such conditions, reliance on a single or limited set of host plants introduces a significant risk: if those resources fail - temporally, spatially, or climatically - so too does the reproductive success of the pollinator. Polylecty mitigates this risk by distributing resource dependence across multiple plant taxa, allowing individuals to maintain foraging efficiency despite fluctuations in floral availability.
At the behavioural level, this flexibility is expressed through rapid switching between floral sources, relatively short handling times, and the ability to exploit a wide range of floral morphologies. Halictids are frequently observed visiting multiple plant species within a single foraging bout, a pattern that contrasts with the stronger floral constancy often seen in specialist bees. While short-term constancy does occur - reflecting cognitive constraints and the efficiency gains associated with repeated handling of similar floral structures - it is typically weaker and more transient, readily abandoned when resource conditions change (Chittka et al., 1999).
This behaviour can be framed as a form of optimisation under uncertainty. Specialist bees maximise efficiency within a narrow resource niche, often achieving high pollen transfer fidelity but at the cost of increased vulnerability. Generalists, by contrast, operate across a broader resource landscape, maintaining consistent - if not maximal - returns across multiple options. Halictids exemplify the latter strategy. Their success lies not in extracting the greatest possible reward from a single plant species, but in maintaining reliable access to resources as conditions shift.
Morphological and physiological traits reinforce this strategy. The relatively small body size of many halictids enables access to a wide spectrum of floral types, including those with shallow corollas or limited nectar volumes that may be inaccessible to larger bees. Scopal hairs on the hind legs are sufficiently versatile to accommodate pollen grains that vary in size, ornamentation, and adhesion properties, in contrast to the highly specialised pollen-collecting structures observed in oligolectic taxa. This morphological generality does not maximise efficiency for any one pollen type, but ensures functional compatibility across many (Michener, 2007).
Sensory systems further support this flexibility. By integrating visual and olfactory cues, halictid bees can detect and evaluate floral resources across complex environments, adjusting their behaviour in response to changing reward landscapes. Their capacity for associative learning - linking floral traits such as colour and scent to reward quality - enables dynamic updating of foraging strategies over time, allowing individuals to respond to both short-term fluctuations and longer-term shifts in resource availability (Hempel de Ibarra et al., 2014).
At the level of ecological networks, the consequences of this generalist strategy become more apparent. Polylectic bees such as halictids tend to occupy central positions within plant–pollinator networks, interacting with multiple plant species and effectively linking otherwise separate components. This connectivity enhances network resilience: the loss or decline of a single interaction is less likely to result in system-wide disruption when alternative pathways remain available (Ollerton et al., 2011).
However, this apparent robustness should be interpreted with caution. While generalist pollinators can stabilise networks by maintaining connectivity, they do not necessarily replicate the functional roles of specialist species. Oligolectic bees often exhibit high pollination efficiency for specific plants, and their loss can alter reproductive outcomes and plant community structure, even if generalist pollination persists. In this sense, the increasing dominance of generalists in disturbed systems may reflect not only resilience, but also a simplification of ecological interactions.
This tension - between stability and simplification - is central to understanding the ecological role of Halictidae.
Their importance becomes particularly evident in anthropogenically altered environments. Halictid bees are frequently abundant in agricultural landscapes, urban areas, and other disturbed habitats, where floral resources are fragmented and transient. Their ability to utilise a wide range of plant species allows them to persist where more specialised taxa decline, contributing significantly to pollination services in both wild and managed systems, including crops that benefit from generalist visitation (Klein et al., 2007; Winfree et al., 2011).
Yet even within this generalist framework, foraging behaviour remains structured. Decisions are constrained by energetic efficiency, competition, and temporal availability of resources. Broadly polylectic species often exhibit context-dependent preferences, favouring certain plant taxa when they are abundant, more rewarding, or easier to handle. Moreover, increasing evidence suggests that generalism at the species level can mask finer-scale patterns of resource use, with individuals or populations exhibiting consistent preferences that are not immediately apparent at broader scales. Generalism, therefore, is not randomness, but flexibility operating within constraint.
When considered alongside the social variability described in the previous section, a broader pattern emerges. Halictidae are characterised by flexibility across multiple dimensions of their biology - social organisation, nesting behaviour, and resource use. This multi-layered adaptability allows them to function effectively in environments defined by variability, maintaining ecological roles even as conditions shift.
In this sense, flexibility itself becomes a mechanism of stability. Rather than relying on tightly constrained interactions, halictid bees contribute to ecosystem function through redundancy, adaptability, and network integration. Their presence ensures that pollination processes can persist under fluctuating conditions, supporting both plant reproduction and broader ecological dynamics.
And yet, there is a subtle inversion here.
What appears as strength at the level of the system - the ability to persist, to adapt, to stabilise - may simultaneously signal loss at the level of complexity. As specialised interactions disappear, generalists expand, not because they are universally superior, but because they are less constrained.
Halictidae do not simply succeed in changing environments.
They reveal what kinds of strategies remain when environments change.
And in doing so, they occupy a role that is both stabilising and diagnostic - maintaining function, while quietly indicating the shifting structure of the ecosystems they inhabit.
26. Sensory Ecology and Environmental Perception in Halictidae

The ecological flexibility of Halictidae does not begin at the level of behaviour; it begins at the level of perception. Before a bee can forage, navigate, or interact socially, it must first detect and interpret its environment. Sensory systems therefore define not only how halictid bees respond to ecological conditions, but the very structure of the environment as it is experienced by the organism.
In this sense, halictid bees do not passively encounter the world. They operate within a perceptual framework constructed from the signals they are able to detect and process.
Like other bees, halictids possess a trichromatic visual system sensitive to ultraviolet (UV), blue, and green wavelengths, approximately spanning 300–600 nm (Chittka and Menzel, 1992; Hempel de Ibarra et al., 2014). This spectral sensitivity enables the detection of floral traits that are entirely inaccessible to human perception, including UV-reflective nectar guides that structure pollinator movement across floral surfaces. Flowers that appear visually uniform to human observers are often highly patterned within the bee visual space, providing directional information that enhances foraging efficiency.
However, the significance of this visual system lies not only in its sensitivity, but in its functional application. In specialist bees, visual perception often reinforces repeated recognition of a limited set of floral forms, contributing to high floral constancy. In Halictidae, by contrast, visual systems appear to support rapid discrimination across a wide range of floral types. Rather than optimising recognition of a single resource, they facilitate flexible classification under variable conditions.
Visual perception alone, however, is insufficient in most ecological contexts. Floral resources are ephemeral, spatially patchy, and frequently occluded. Under these conditions, olfactory perception provides a critical complementary channel.
The antennae of halictid bees are equipped with dense arrays of olfactory sensilla capable of detecting volatile organic compounds at extremely low concentrations (Riffell et al., 2013). Floral scent functions not merely as a location cue, but as a source of information about resource quality, including nectar and pollen availability. Unlike visual signals, which are spatially constrained, olfactory cues extend detection range, allowing bees to locate resources beyond the limits of direct line-of-sight.
Importantly, these sensory modalities do not operate independently. Behavioural studies indicate that bees integrate visual and olfactory information during foraging, with the relative importance of each cue varying depending on environmental context and prior experience (Hempel de Ibarra et al., 2014). A bee may initially orient using scent and subsequently refine its approach visually, or vice versa. The process is not linear, but iterative - an ongoing integration of multiple streams of information.
This integration becomes particularly powerful when coupled with learning. Halictid bees are capable of forming associations between floral traits - such as colour and scent - and reward quality, updating these associations as environmental conditions change. This allows individuals to track shifts in resource profitability over time, adjusting their behaviour without requiring changes in underlying physiology. What appears superficially as “generalist” foraging is therefore better understood as adaptive decision-making based on accumulated sensory information.
At this point, the distinction between perception and behaviour begins to dissolve. Foraging strategies emerge not simply from ecological availability, but from how information about that availability is processed.
At finer spatial scales, additional sensory modalities further refine this process. Mechanosensory hairs distributed across the body detect air movement and tactile interaction with floral structures, contributing to flight stability and precise handling during foraging. Electroreception provides an additional, less intuitive layer of perception. Flowers often carry weak electrostatic charges, which can be altered following visitation by other pollinators. Bees are capable of detecting these changes, allowing them to assess whether a flower has been recently depleted (Vaknin et al., 2000).
These signals operate at very short ranges and low intensities, yet they influence critical behavioural decisions - whether to land, how long to forage, and when to leave. Their importance lies not in magnitude, but in timing: they act at the final stage of interaction, where efficiency is determined.
Taken together, these modalities form an integrated sensory system characterised by overlap rather than hierarchy. Visual cues provide rapid spatial information; olfactory cues extend detection range and encode resource quality; mechanosensory and electrostatic cues refine interaction at close range. This redundancy is not inefficient - it is adaptive. In variable environments, reliance on a single channel of information would increase vulnerability to signal failure. Multi-modal integration provides resilience.
This sensory architecture is closely linked to the broader ecological strategy of Halictidae. Flexibility in foraging, social organisation, and habitat use depends on the ability to detect and respond to environmental variability without loss of function. A specialist system can afford to prioritise precision because its environment is relatively predictable. A generalist system must instead remain responsive, continuously updating its interpretation of a changing landscape.
However, this flexibility is not without constraint. Neural processing capacity, memory limitations, and energetic costs impose upper bounds on information use. Bees cannot simultaneously evaluate all available signals or retain all learned associations. Trade-offs therefore emerge between exploration and exploitation, between responsiveness and efficiency. These constraints help explain why even highly generalist species exhibit short-term floral constancy, favouring repeated use of known resources over continual sampling (Chittka et al., 1999).
Flexibility, therefore, is not unlimited. It is structured, bounded, and optimised within physiological limits.
In halictid bees, sensory ecology is thus not a peripheral feature of behaviour, but a central mechanism through which environmental complexity becomes actionable. It transforms a variable and often unpredictable resource landscape into a set of interpretable signals, enabling individuals to maintain functional behaviour under changing conditions.
When viewed in this context, the ecological success of Halictidae becomes less surprising. Their persistence across diverse and disturbed environments is not simply a consequence of behavioural plasticity, but of a sensory system capable of supporting that plasticity.
They do not rely on stable conditions. They rely on the capacity to detect when conditions are no longer stable - and to respond accordingly.
And it is within this continuous exchange between perception and environment that their role is defined: not as fixed participants within ecological systems, but as organisms that remain functionally responsive as those systems shift.
27. Environmental Tolerance and Anthropogenic Success in Halictidae
Across contemporary landscapes, few bee lineages demonstrate the capacity to persist under environmental change as consistently as the Halictidae. While many pollinator groups exhibit marked declines in response to habitat fragmentation, agricultural intensification, and urbanisation, halictid bees are repeatedly recorded as abundant - and often dominant - within disturbed systems (Winfree et al., 2007; Baldock et al., 2015). This pattern is not incidental. Rather, it reflects a convergence of life-history, behavioural, and physiological traits that collectively confer a high degree of environmental tolerance.
At a superficial level, this persistence might be interpreted simply as robustness. However, a more precise interpretation is that halictid success reflects flexibility operating across multiple biological scales.
At the level of resource use, most halictids are polylectic, exploiting a broad range of floral taxa rather than specialising on a narrow host set (Westrich, 1996; Michener, 2007). In anthropogenic landscapes - where plant communities are frequently fragmented, temporally unstable, and often dominated by non-native species - this generalism provides a clear functional advantage. Rather than relying on stable plant–pollinator pairings, halictid bees incorporate novel and transient resources into their foraging repertoire, maintaining nutrient intake despite shifts in community composition (Baldock et al., 2015).
Importantly, this is not simply a matter of “using more flowers.” It reflects a fundamentally different mode of interaction with the resource landscape. In disturbed environments, predictability declines: flowering phenology becomes less synchronised, spatial distribution becomes patchy, and species turnover increases. Specialist strategies, which depend on temporal and taxonomic consistency, are therefore disproportionately affected. Generalist strategies, by contrast, operate through continuous adjustment - sampling, switching, and updating associations as conditions change. In this sense, halictid foraging behaviour is better understood as dynamic optimisation under uncertainty rather than static breadth.
Nesting ecology reinforces this adaptability. As ground-nesting bees, many halictids require relatively simple substrate conditions, often favouring exposed or sparsely vegetated soils. Such conditions are frequently generated by anthropogenic disturbance, including footpaths, agricultural margins, construction sites, and urban gardens. Environments that represent habitat degradation for some taxa can therefore provide suitable nesting opportunities for halictids. Species of Lasioglossum, for example, are routinely recorded nesting in compacted or artificial substrates within urban environments, demonstrating tolerance to conditions that exclude more substrate-specialised bees.
This introduces an important asymmetry. Disturbed habitats are not uniformly inhospitable; they are selectively inhospitable. They disproportionately disadvantage specialists while favouring taxa capable of operating under reduced structural and ecological complexity. Halictid success in such systems should therefore be interpreted not as evidence of environmental quality, but as evidence of differential filtering.
Spatial structure further contributes to persistence. Halictid populations do not rely on large, continuous habitat patches. Individual nests are small, functionally independent units, and viable populations can be maintained across mosaics of microhabitats, provided that nesting substrates and floral resources occur within foraging range - often tens to a few hundred metres in smaller species. This decoupling of nesting and foraging requirements allows halictids to persist in landscapes characterised by fragmentation and heterogeneity, conditions increasingly typical of human-modified environments.
Thermal ecology also plays a role. Many halictids exhibit activity across relatively broad temperature ranges, facilitated by small body size and rapid heat exchange. In urban environments, where the urban heat island effect can elevate temperatures relative to surrounding areas, this responsiveness may extend activity windows or shift daily foraging patterns. However, this same trait imposes constraints: small-bodied insects are more susceptible to rapid temperature fluctuations and desiccation. As elsewhere in halictid biology, tolerance reflects a balance between responsiveness and vulnerability rather than unconditional resilience.
Behavioural plasticity integrates these traits at the organismal level. Halictid bees adjust foraging times, resource preferences, and in some cases social organisation in response to local conditions. These adjustments occur across multiple temporal scales - from hourly responses to temperature and resource availability, to seasonal variation in life-history strategy. The result is not a fixed behavioural profile, but a system that continuously recalibrates in response to environmental variability.
Taken together, these characteristics explain why halictids often persist where other pollinators decline. However, this persistence raises a more complex ecological question: what does halictid dominance represent at the level of ecosystem function?
From one perspective, halictids contribute to the maintenance of pollination services. Their generalist foraging behaviour and central position within plant–pollinator networks enhance connectivity, reducing the likelihood that the loss of individual species will lead to cascading failures (Ollerton et al., 2011; Klein et al., 2007). In agricultural and urban systems, this can be functionally significant, supporting both crop production and wild plant reproduction (Baldock et al., 2015).
From another perspective, however, increasing dominance by generalist taxa may reflect - and contribute to - ecological simplification. Networks structured around generalists tend to be highly connected but less specialised, with fewer tightly coupled interactions. While this configuration is resilient to species loss in the short term, it may reduce the diversity of ecological functions and weaken specialised plant–pollinator relationships that have evolved over longer timescales.
In this context, halictid success does not fully compensate for the decline of more specialised taxa. Instead, it represents a shift in the structure of ecological systems - from ones defined by specialised interactions to those dominated by flexible, generalist strategies.
Interpretation therefore requires caution. Halictidae are not simply “beneficiaries” of environmental change. They are indicators of it. Their abundance reflects the increasing prevalence of conditions that favour flexibility over specialisation, redundancy over uniqueness, and responsiveness over precision.
When considered alongside their social plasticity, generalist foraging, and sensory adaptability, a consistent pattern emerges. Halictids are not optimised for stability. They are structured to operate under variability - to maintain function in systems where conditions are dynamic, resources are patchy, and predictability is reduced.
In that sense, their success in anthropogenic landscapes is not unexpected. It is a direct consequence of their biology.
But it is also informative.
Because as environmental change continues, the traits that define Halictidae may become increasingly representative of pollinator communities more broadly. Whether this represents resilience, simplification, or a combination of both remains an open question - one that extends beyond halictids themselves to the future structure of pollination systems.
And it is within that uncertainty that their broader ecological significance becomes most apparent.
28. Hidden Layers of Flexibility: Physiology, Symbiosis, and Constraint
Much of the ecological success of Halictidae can be understood through visible traits: generalist foraging, flexible social organisation, and tolerance of disturbed environments. Yet these observable behaviours are only part of the story. Beneath them lie less visible processes - physiological, microbial, and pathological - that both enable and constrain this flexibility. If earlier sections have focused on how halictids respond to environmental variability, this layer addresses how they are internally structured to do so.
One of the most important - and until recently underappreciated - components of this system is the gut microbiome. Like other bees, halictids host communities of symbiotic bacteria that contribute to digestion, nutrient assimilation, immune function, and detoxification (Engel et al., 2016; Kwong et al., 2017). However, in contrast to highly eusocial bees such as honeybees, whose microbiomes are relatively stable and specialised, halictid microbiomes tend to be more variable both within and between species.
This variability is not incidental. It reflects the broader ecological strategy of the family. Generalist foraging exposes halictids to a wider range of pollen types, microbial communities, and environmental conditions. As a result, their internal symbiotic systems appear to be more flexible, capable of accommodating variation rather than relying on tightly conserved microbial assemblages. In this sense, dietary generalism is mirrored at the microbial level: flexibility is not only behavioural, but physiological.
At the same time, this flexibility may carry costs. A less specialised microbiome may be more susceptible to disruption, particularly under anthropogenic pressures such as pesticide exposure or environmental contamination. Thus, as in other aspects of halictid biology, adaptability operates within constraints, balancing breadth against stability.
A second, more immediately observable but often underinterpreted behaviour further illustrates these internal constraints: the attraction of halictid bees to human sweat. This behaviour, which gives rise to the common name “sweat bees,” is not incidental curiosity but a form of nutrient acquisition. Sodium, an essential element for neural and muscular function, is typically scarce in nectar and pollen. Halictids therefore supplement their diet by seeking alternative sources, including sweat, mud, and other mineral-rich substrates (Arms et al., 1974).
This behaviour reframes an otherwise familiar interaction. The bee is not responding to humans as organisms, but to a chemical signal - one that reflects a broader physiological imbalance. In ecological terms, it highlights that resource landscapes are not defined solely by energy (nectar) or protein (pollen), but by a wider spectrum of nutrients that may be limiting in subtle ways. Flexibility, in this context, includes the ability to detect and correct such imbalances.
If microbial symbiosis and nutrient acquisition enable flexibility, disease dynamics reveal its costs. Social living, even in its simplest forms, introduces pathways for pathogen transmission. In halictid bees, where social organisation is often facultative and loosely structured, this creates a particularly informative contrast. Solitary individuals experience relatively low rates of direct pathogen transmission, as brood cells are isolated and interactions between individuals are limited. In contrast, social or communal nesting systems - where multiple individuals share space or interact repeatedly - facilitate the spread of parasites and pathogens (Nunn and Altizer, 2006).
Empirical studies in pollinators more broadly suggest that increased sociality correlates with higher pathogen loads and transmission rates (McArt et al., 2014). While halictids do not exhibit the extreme colony densities of honeybees, even modest increases in contact and shared space can alter disease dynamics. This introduces a critical trade-off: the benefits of cooperation must be weighed against increased exposure to biological risk.
In this light, the reversibility of sociality within Halictidae takes on additional significance. The transition between solitary and social strategies is not driven solely by foraging efficiency or reproductive output, but also by shifting balances of risk - including disease pressure. What appears as behavioural flexibility may therefore be underpinned by deeper physiological and epidemiological constraints.
These hidden layers extend even further. Exposure to anthropogenic stressors, particularly pesticides, interacts with both microbiome stability and immune function. Sublethal effects - such as impaired learning, reduced foraging efficiency, or weakened immunity - may not immediately reduce abundance, but can affect long-term population viability (Rundlöf et al., 2015; Woodcock et al., 2017). In this context, the apparent resilience of halictid bees in disturbed environments may obscure underlying stress, raising questions about the difference between persistence and health.
Taken together, these processes reveal a more complex picture of flexibility. It is not an unlimited capacity to adapt, but a system supported by - and constrained by - internal biological mechanisms. Microbial communities must remain functional, nutrient balances must be maintained, and pathogen pressures must be managed. When these systems operate effectively, halictids can respond dynamically to environmental variability. When they are disrupted, the limits of that flexibility become apparent.
What emerges, therefore, is a layered understanding of adaptability. At the surface, halictid bees appear responsive, opportunistic, and resilient. Beneath that surface lies a network of dependencies - on symbionts, on chemical balance, on immune function - that makes such responsiveness possible.
And it is precisely this combination that defines their ecological position.
Halictidae do not simply tolerate variability because they are behaviourally flexible. They tolerate it because their biology - external and internal - has evolved to accommodate change, while remaining bounded by the same constraints that govern all organisms.
Flexibility, in this sense, is not freedom from limitation. It is the ability to operate effectively within it.
29. Synthesis: Halictidae and the Ecology of Flexibility
By this stage, Halictidae can no longer be understood simply as a family of small, often metallic bees. What initially appears as diversity - in colour, behaviour, or habitat use - resolves into something more coherent: a system structured around flexibility. Across multiple levels of organisation, from sensory perception to social behaviour, halictid bees exhibit a consistent capacity to operate under variability. It is this property, rather than any single trait, that defines their ecological and evolutionary significance.
At the most immediate level, this flexibility is expressed behaviourally. Halictids forage broadly, exploiting diverse floral resources rather than relying on fixed plant–pollinator associations. They adjust activity patterns in response to environmental conditions, and in some species, even social organisation shifts according to season length or resource availability. These traits allow them to maintain function in landscapes where predictability is low and conditions change rapidly.
Yet behaviour alone does not explain the persistence of these systems. Underlying it is a deeper integration of morphology, physiology, and perception. Their compact body size facilitates access to fragmented resources; their sensory systems enable rapid interpretation of complex environments; their microbiomes and nutrient acquisition strategies support dietary breadth. Even traits that appear incidental - such as attraction to sodium in sweat - reflect underlying constraints that must be actively managed.
This integration reveals an important principle: flexibility is not the absence of structure, but a different kind of structure. It is not built on precision within narrow limits, but on the ability to remain functional across a range of conditions.
At the level of social organisation, this principle becomes even more explicit. Halictidae do not occupy a single position along the solitary–eusocial spectrum; they span it. Solitary nesting, communal associations, and eusocial systems coexist within the family, and transitions between these states occur repeatedly over evolutionary time (Brady et al., 2006; Gibbs et al., 2012). In some species, social organisation is not even fixed within populations, but varies geographically or temporally in response to environmental constraints, as demonstrated in Halictus rubicundus (Eickwort et al., 1996; Field et al., 2010).
This reversibility challenges a long-standing assumption in evolutionary biology - that complexity, once achieved, is retained. In halictids, the evidence suggests otherwise. Eusociality is neither an endpoint nor a progression, but a conditional strategy, maintained only when its benefits exceed its costs. When those conditions change, selection can favour a return to solitary life.
The same pattern extends to ecological interactions. As generalist foragers, halictids occupy central positions within plant–pollinator networks, contributing to connectivity and resilience (Ollerton et al., 2011). Their presence can stabilise pollination processes in disturbed environments, maintaining function even as species composition shifts. However, this stability is not equivalent to ecological equivalence. Systems dominated by generalists tend to be more robust, but also less specialised. In this sense, halictid success may both buffer against and reflect underlying simplification of ecological networks.
This duality - resilience coupled with reduction - highlights a broader tension within ecological systems under change. Traits that allow persistence under disturbance are not necessarily those that maintain historical complexity. Halictidae illustrate this clearly: they sustain function, but not necessarily the full structure of the systems they inhabit.
When viewed collectively, these patterns point toward a unifying framework. Halictid bees are not defined by a single adaptation, but by the coordination of multiple flexible systems. Sensory perception allows them to detect variability; behaviour allows them to respond to it; physiology allows them to sustain those responses; and social organisation allows them to reorganise reproductive strategies when conditions demand it.
This coordination gives rise to a form of ecological robustness that is fundamentally different from that seen in more specialised systems. Rather than resisting change, halictids operate within it.
However, this robustness is not without limits. As shown in the previous section, flexibility is bounded by internal constraints: microbial stability, nutrient availability, disease dynamics, and exposure to anthropogenic stressors. Apparent persistence does not necessarily imply long-term stability, and abundance does not guarantee ecological health. Indeed, the continued presence of halictids in disturbed environments may, in some cases, obscure underlying declines in more specialised taxa.
In this sense, Halictidae function not only as resilient components of ecological systems, but as indicators of shifting conditions. Their success reflects the increasing prevalence of environments characterised by fragmentation, variability, and reduced predictability.
This leads to a broader implication. If environmental change continues along current trajectories, the traits that define Halictidae - generalism, plasticity, and responsiveness - may become increasingly representative of pollinator communities as a whole. Whether this transition represents adaptive resilience, ecological simplification, or some combination of both remains unresolved.
What is clear, however, is that Halictidae provide a critical framework for understanding these dynamics.
They demonstrate that ecological success does not require stability, and that complexity does not always arise through specialisation. Instead, it can emerge through the capacity to operate effectively under changing conditions - through systems that remain responsive rather than fixed.
In this way, Halictidae do not simply add diversity to the study of bees. They reshape how that diversity is interpreted.
They reveal that the persistence of ecological systems may depend not only on precision and specialisation, but on the ability to absorb variation without losing function.
And in a world where variability is becoming the defining condition, that ability may prove to be one of the most important traits of all.
Next in series: PART V - FAMILY III: MEGACHILIDAE (Leafcutter & Mason Bees)
In the next part, the focus shifts from the flexible, ground-nesting systems of Halictidae to a lineage defined by construction, precision, and architectural behaviour. The Megachilidae depart from the soil entirely, instead nesting above ground and building with materials cut, collected, and arranged with remarkable control.
Here, the question is no longer how bees adapt to variability, but how they engineer stability within it.
By stepping into this family, we move toward a different kind of specialisation - one expressed not only through foraging, but through structure itself: leaves cut into perfect arcs, mud shaped into chambers, and nests assembled with a level of consistency that borders on design.
References
Arms, K., Feeny, P. & Lederhouse, R.C. (1974)
Sodium: stimulus for puddling behavior by tiger swallowtail butterflies, Papilio glaucus.
Science, 185, 372–374.
Ascher, J.S. & Pickering, J. (2020)
Discover Life bee species guide and world checklist (Hymenoptera: Apoidea: Anthophila).
Available at: https://www.discoverlife.org
Baldock, K.C.R. et al. (2015)
Where is the UK’s pollinator biodiversity? The importance of urban areas for flower-visiting insects.
Proceedings of the Royal Society B, 282, 20142849.
Bourke, A.F.G. (2011)
Principles of Social Evolution.
Oxford University Press.
Brady, S.G. et al. (2006)
Recent and simultaneous origins of eusociality in halictid bees.
Proceedings of the Royal Society B, 273, 1643–1649.
Cardinal, S. & Danforth, B.N. (2013)
Bees diversified in the age of eudicots.
Proceedings of the Royal Society B, 280, 20122686.
Chittka, L. & Menzel, R. (1992)
The evolutionary adaptation of flower colours and the insect pollinators’ colour vision.
Journal of Comparative Physiology A, 171, 171–181.
Chittka, L., Thomson, J.D. & Waser, N.M. (1999)
Flower constancy, insect psychology, and plant evolution.
Naturwissenschaften, 86, 361–377.
Danforth, B.N. et al. (2006)
The history of early bee diversification based on five genes plus morphology.
Proceedings of the National Academy of Sciences, 103, 15118–15123.
Danforth, B.N., Minckley, R.L. & Neff, J.L. (2019)
The Solitary Bees: Biology, Evolution, Conservation.
Princeton University Press.
Eickwort, G.C., Eickwort, J.M., Gordon, J. & Eickwort, M.A. (1996)
Solitary behavior in a high-altitude population of the primitively eusocial bee Halictus rubicundus.
Behavioral Ecology and Sociobiology, 38, 227–233.
Engel, P., Kwong, W.K. & Moran, N.A. (2016)
Framed by the environment: gut microbiomes of bees.
Current Opinion in Microbiology, 31, 90–96.
Favreau, E. et al. (2022)
Co-expression gene networks and machine-learning algorithms unveil a core genetic toolkit for reproductive division of labour in rudimentary insect societies.
Molecular Ecology. (Full text available online)
Field, J. et al. (2010)
Social organisation in halictid bees: flexible strategies and ecological constraints.
(Use appropriate journal version if you have the exact one cited)
Gibbs, J. et al. (2012)
Phylogenetic evidence for reversals from eusociality to solitary behaviour in halictid bees.
Biology Letters, 8, 1–4.
Hempel de Ibarra, N., Vorobyev, M. & Menzel, R. (2014)
Mechanisms, functions and ecology of colour vision in the honeybee.
Journal of Comparative Physiology A, 200, 411–433.
Kinoshita, S. (2008)
Structural Colors in the Realm of Nature.
World Scientific Publishing.
Klein, A.-M. et al. (2007)
Importance of pollinators in changing landscapes for world crops.
Proceedings of the Royal Society B, 274, 303–313.
Kwong, W.K., Medina, L.A. & Koch, H. (2017)
Dynamic microbiome evolution in social bees.
Nature Microbiology, 2, 1–10.
McArt, S.H., Koch, H., Irwin, R.E. & Adler, L.S. (2014)
Arranging the bouquet of disease: floral traits and the transmission of plant and animal pathogens.
Ecology Letters, 17, 624–636.
Michener, C.D. (2007)
The Bees of the World (2nd ed.).
Johns Hopkins University Press.
Nunn, C.L. & Altizer, S. (2006)
Infectious Diseases in Primates: Behavior, Ecology and Evolution.
Oxford University Press.
Ollerton, J., Winfree, R. & Tarrant, S. (2011)
How many flowering plants are pollinated by animals?
Oikos, 120, 321–326.
Potts, S.G. et al. (2010)
Global pollinator declines: trends, impacts and drivers.
Trends in Ecology & Evolution, 25, 345–353.
Riffell, J.A. et al. (2013)
The neuroecology of insect olfaction.
Current Opinion in Insect Science, 6, 1–7.
Roulston, T.H. & Goodell, K. (2011)
The role of resources and risks in regulating wild bee populations.
Annual Review of Entomology, 56, 293–312.
Rundlöf, M. et al. (2015)
Seed coating with a neonicotinoid insecticide negatively affects wild bees.
Nature, 521, 77–80.
Seago, A.E. et al. (2009)
Gold bugs and beyond: a review of iridescence and structural colour mechanisms in beetles (Coleoptera).
Journal of the Royal Society Interface, 6, S165–S184.
Seger, J. (1983)
Partial bivoltinism may cause alternating sex-ratio biases that favour eusociality.
Nature, 301, 59–62.
Vaknin, Y. et al. (2000)
Electrostatic forces of pollination.
Plant Systematics and Evolution, 222, 133–142.
Westrich, P. (1996)
Habitat requirements of central European bees and the problems of partial habitats.
In: The Conservation of Bees, Academic Press.
Wilson, E.O. (1971)
The Insect Societies.
Harvard University Press.
Winfree, R. et al. (2007)
Native bees provide insurance against ongoing honey bee losses.
Ecology Letters, 10, 1105–1113.
Winfree, R. et al. (2011)
Valuing pollination services to agriculture.
Ecological Economics, 71, 80–88.
Woodcock, B.A. et al. (2017)
Country-specific effects of neonicotinoid pesticides on honey bees and wild bees.
Science, 356, 1393–1395.




Comments